About ViraChem
An EU-based platform coordinating peptide manufacturing and sourcing for research and preclinical development.
Vision
To be the leading global platform for the scientific industry through the integration of new technologies to provide the best service.
Our values
- Honest.
- Flexibility.
- Commitment.
ViraChem operates as a layer coordinating sourcing, synthesis, purification, and downstream processing of high-purity peptides and fine chemicals.
Through established manufacturing partnerships in Poland, we support research and preclinical workflows with services including peptide synthesis, lyophilization, and fill & finish.

Who we support
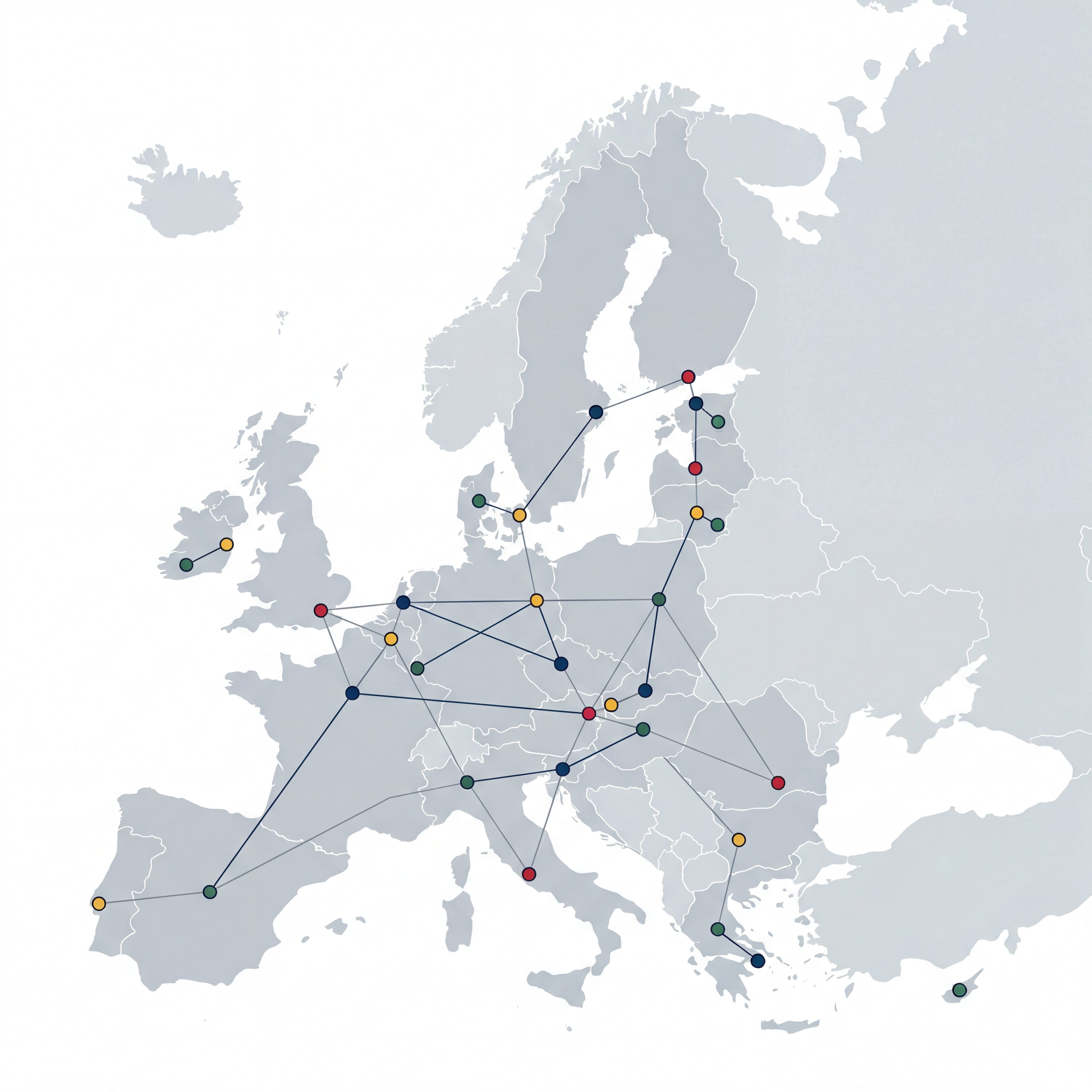
European network
Find us at our EU offices and manufacturing sites — contact us for coordination, quotes, and technical support for research and preclinical work.
Our locations
What we offer
EU service models for research and preclinical supply.
Our position in the Industry Ecosystem
Our role is to simplify the complex, mitigate risk, and ensure that every gram of product meets the exact specifications required by your industry—while keeping coordination responsive and costs predictable. ViraChem sits between upstream manufacturers and downstream users, enabling:
- Secure access to qualified peptide raw materials
- Coordinated EU-based synthesis and processing
- Smooth transition from research to high-volume programs.
- Documentation packages aligned to agreed specifications—for research and preclinical programs.
- Faster quotes, milestones, and handoffs through a single coordination layer
- Competitive, transparent pricing aligned to your volume and specifications
This positioning allows partners to scale without supply chain fragmentation or regulatory ambiguity—with shorter cycle times and stronger cost control at every stage.

Contribute to scientific progress by providing an innovative and quality service that connects the needs of laboratories and manufacturers of chemical and biological products.
Why partners work with us
EU-coordinated manufacturing, documented handoffs, and one point of contact—so your team gets traceability and continuity, not guesswork.
Built for Research-Grade Peptide Supply
We deliver more than sourcing: technical depth, transparent milestones, and coordination built for research-grade peptide programs.

We excel in key areas, showcasing our services.
Research raw powder
Upstream access to high-purity research-grade peptide raw powder
Finished RUO Formats
Short-term access for early-stage or time-sensitive research programs.
Custom Synthesis & Sourcing
Core engagement model for institutional partners.
Fill & Finish Services
Processing and labeling for clients managing their own manufacturing workflows.
Cosmetic White-Label Formats
Licensed peptide-based cosmetic products (creams, serums, gels) for clinics, brands, or labs—with ViraChem-sourced ingredients and optional white-label branding.
Meet the founders

ViraChem was established to offer European labs and industrial partners reliable access to peptide manufacturing and research-grade raw materials, minimizing supply chain risks and regulatory issues.

Radoslav oversees partner acquisition and manufacturing coordination at ViraChem's EU network. He works with clinics, labs, and institutions to build supply relationships, ensuring technical feasibility and production alignment.